Note: This is the first of many posts for our new website designed to highlight the role of Earth Sciences within UConn Nation. The target audience consists of students, faculty, staff, and administrators interested in how the earth works, what its history has been, and why this matters.
As our climates change, water is increasingly finding its way into our political and strategic discourse, with special attention given to water scarcity during droughts and water excesses during storm deluges. Yet seldom do I hear these critical issues linked to earth sciences. Why? This oversight derives from general ignorance of how the earth works, what it’s history has been, and why this matters as a consequence of K-12 curriculum. It is our discipline, and no other at UConn, that provides the planetary context that supports environmental sciences and environmental studies.
By water, I mean all three material phases of this life-giving substance — the melting of solid water from snowpacks glaciers, the fluid filling of our ocean basins, and the vapor transformations responsible for creating droughts and wet hurricanes. Indeed, H2O is a versatile molecule that manifests as a mineral when cold enough and as a melt in Earth’s ambient climes.
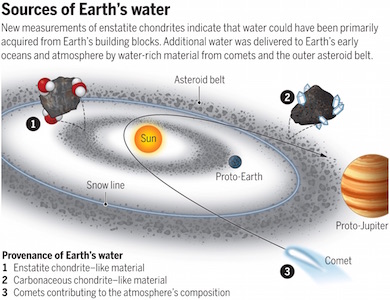
Thanks to a recent Research Report in Science, by Piani et al. (DOI: 10.1126/science.aba1948) and to an accompanying Insight Perspective by Anne E. Peslier (DOI: 10.1126/science.abc1338), we finally figured out where most of Earth’s water actually came from. The answer? A mineral familiar to every student who’s taken any one of our introductory earth science courses: pyroxene, a chief component of the familiar crustal rock basalt, and of the less familiar rocks of the mantle. More specifically, water came from a magnesium-rich pyroxene known as enstatite (MgSiO3).
The origin of Earth’s water has long been a matter of considerable debate. We’ve known for decades that the main building blocks for the inner planets of our solar system –Mercury, Venus, Earth, and Mars– were primitive solid aggregates, called chondrites, that were rich in enstatite. Previously, we’ve thought that this type of chondrite was too dry to be the source of Earth’s water. The reigning counter-hypothesis has long been that the bulk of Earth’s water came from cabonaceous chondrites and ice particles drawn in from the colder, outer reaches of our solar system very late in Earth’s formation. Now we know from this recent careful isotopic work that there’s enough water in enstatite chondrites to fill all of earth’s ocean basins several times over with the liquid that keeps our planet blue. This happened in two stages: initially when Earth melted and steamed that water into the Hadean atmosphere; and later when earth cooled enough to allow that steam to condense and fall as rain.
For many years, I’ve offered to students, public audiences, and readers a five-word, 1-minute summary of Earth history. In chronological sequence: “Rock. Air. Water. Life. Intelligence?” This turns out to be truer than I previously thought. Reading this story backwards, our unique intelligence came from life in general, which came from liquid water, which came from the steamy atmosphere, which came from rock. At UConn, the Department of Earth Sciences works on all these pieces of Earth’s magnificent story.